Latisse
Conveniently located to serve the areas of Sunset Beach, Newport Beach, Redondo Beach, Orange, Dana Point and Orange County

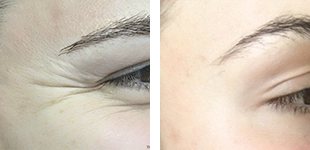
DermFx is proud to provide our patients with LATISSE® Solution treatment. LATISSE® Solution is the first prescription treatment approved by the FDA for the treatment of small or thin eyelash growth. When applied regularly on a daily basis, the LATISSE® Solution product results in the growth of significantly longer, fuller, and darker upper eyelid lashes.
The product was discovered by researchers who were working on another eye care product, a glaucoma medication. But they quickly realized its potential for eyelash growth. The mechanism of the product’s active ingredient, called bimatoprost, is not known precisely, but researchers believe the results are due to an increase in the percentage of lash development during the lash growth phase. About 80% of Latisse users report seeing a significant increase in the growth and fullness of lashes.
Contents
About
LATISSE® Solution is a prescription product which you apply yourself at home. Prior to applying the product, you should wash your face and remove any makeup or creams. Contact lens should also be removed. Prepare the special applicator by placing a drop of solution on the applicator and apply it to the base of the upper eyelid where the lash base meets the skin of the eyelid. Any excess solution should be blotted off with a dry tissue. Use a fresh applicator for each lid – never reuse an applicator. Some individuals have reported temporary itching or slight redness after application, but these conditions usually resolve once the body becomes accustomed to the product. LATISSE® Solution is meant to be applied to the lid, not to the eye itself; however, if a small amount of the solution enters the eye, it will not harm the eye.
Candidates
Any individual who wants to experience fuller, thicker, darker upper lashes is a potential candidate for the LATISSE® Solution program. In research, even those with sensitive eyes were able to use the product successfully. However, there are some contraindications for its use. Any individual on antibiotics, heart medication, or other drugs, as well as those with neurological and certain other diseases, and those with severe allergies should discuss these conditions prior to beginning the program. In addition, pregnant women, as well as those with skin infections or other conditions on the upper eyelids, are not good candidates.
Treatment
The effects of LATISSE® Solution remain only as long as the product is used. It must be applied every day in the initial 16 weeks of treatment in order to see the complete and optimal lash growth effects; however, many patients begin a maintenance phase after those initial 4 months, applying the product every 2 to 3 days to maintain the effect. Individuals should understand, though, that currently recommended dosing instructions advise the daily use of LATISSE, even after the initial 16-week period. The product is meant to be used continuously, and once treatment stops, you can expect your lashes to return to their pretreatment appearance at 4 to 6 weeks after ending treatment.
FAQ
Is the Lash Growth Permanent?
The lashes which grow as a result of the LATISSE® Solution program will last for the natural duration of the eyelash growth cycle, which is usually 1 to 2 months.
What Are the Advantages of LATISSE® Over Other Options?
LATISSE® Solution is currently the only FDA-approved prescription product for the growth and development of thicker, fuller, darker lashes; therefore, current alternatives include the use of false eyelashes or thick mascara. LATISSE® Solution offers fuller, thicker, darker lashes without makeup or prosthetics, and actually causes additional lashes to grow.
Is LATISSE® Solution Approved for Use in the USA?
Yes, LATISSE® Solution is approved by the FDA for lash growth application in the United States.
 DermFx
DermFx